Schrodinger's Contribution to the Atomic Theory
Schrodinger's Wave Equation (1926)
In 1926, Erwin Schrodinger created an equation called Schrodinger's Wave Equation. Schrodinger's Equation predicts the positions of a particle at a particular time. Particles (eg: electrons) behave like waves, meaning they don't travel in a defined orbits or lines. Nobody can tell exactly where a particle will be until they us a device to observe it. Theoretically, scientists can predict where a particle might be if they create a wave equation. However, nobody created such an equation until Schrodinger came along. Using wave mechanics and the fact that particles behave like waves, Schrodinger created an equation that would figure out the probability of a particle being at a particular spot at a particular time. The equation tell you where a particle might be and what the chances are of the particle being there, but it does not give a specific place a particle will be. This equation became the basis for many other studies in quantum mechanics and wave mechanics.
In 1926, Erwin Schrodinger created an equation called Schrodinger's Wave Equation. Schrodinger's Equation predicts the positions of a particle at a particular time. Particles (eg: electrons) behave like waves, meaning they don't travel in a defined orbits or lines. Nobody can tell exactly where a particle will be until they us a device to observe it. Theoretically, scientists can predict where a particle might be if they create a wave equation. However, nobody created such an equation until Schrodinger came along. Using wave mechanics and the fact that particles behave like waves, Schrodinger created an equation that would figure out the probability of a particle being at a particular spot at a particular time. The equation tell you where a particle might be and what the chances are of the particle being there, but it does not give a specific place a particle will be. This equation became the basis for many other studies in quantum mechanics and wave mechanics.
 Bohr's model
Bohr's model
Quatum Mechanical Model (1926)
Before the quantum mechanical model was developed by Schrodinger, the accepted model for atoms was the Bohrs model (Obviously named after Neils Bohr). You may have seen this:
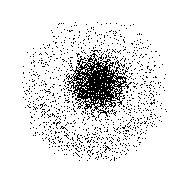
It was based on the theory that electrons in an atom occupy certain paths (orbits) around the nucleus. Schrodinger proposed a new model that was more vague than Bohr's, but also more realistic. Because electrons act like waves, we can not tell exactly where one will be. However, it is possible to calculate the probability of an electron occupying a certain orbit using Schrodinger's wave equation. That was what he (Schrodinger) did. Unlike Bohr's model, Schrodinger's quantum mechanical model of the atom does not define the exact path of the electron. Rather, it predicts the positions of the electrons and the probability for the electron to be in that position. Schrodinger's model is often represented as a cloud surrounding a nucleus:
Before the quantum mechanical model was developed by Schrodinger, the accepted model for atoms was the Bohrs model (Obviously named after Neils Bohr). You may have seen this:
It was based on the theory that electrons in an atom occupy certain paths (orbits) around the nucleus. Schrodinger proposed a new model that was more vague than Bohr's, but also more realistic. Because electrons act like waves, we can not tell exactly where one will be. However, it is possible to calculate the probability of an electron occupying a certain orbit using Schrodinger's wave equation. That was what he (Schrodinger) did. Unlike Bohr's model, Schrodinger's quantum mechanical model of the atom does not define the exact path of the electron. Rather, it predicts the positions of the electrons and the probability for the electron to be in that position. Schrodinger's model is often represented as a cloud surrounding a nucleus:
(The nucleus is at the center, though you probably can't tell from this illustration)
The densest part of the cloud (the part with the most dots) is where electrons are most likely going to be when you measure them, and the less dense parts are where they are less likely be be. This new model of the atom correctly represented the fact that electrons do not have set paths and can not be accurately predicted.
The densest part of the cloud (the part with the most dots) is where electrons are most likely going to be when you measure them, and the less dense parts are where they are less likely be be. This new model of the atom correctly represented the fact that electrons do not have set paths and can not be accurately predicted.